![SOLVED: Determine whether each of the molecules below is polar or nonpolar: BrCl3 nonpolar CS2 [Select ] XeO3 nonpolar polar SO3 SOLVED: Determine whether each of the molecules below is polar or nonpolar: BrCl3 nonpolar CS2 [Select ] XeO3 nonpolar polar SO3](https://cdn.numerade.com/ask_previews/3a82ef-edc8-1cc5-2c82-cb8fe3b6e12_large.jpg)
SOLVED: Determine whether each of the molecules below is polar or nonpolar: BrCl3 nonpolar CS2 [Select ] XeO3 nonpolar polar SO3

Which of the following molecules is polar? (a) NBr_3 (b) CS_2 (c) NH_4^+ (d) CH_4 (e) PCl_5 | Homework.Study.com

OneClass: b. NF3 e SCl2 C. BraN 2. Determine whether each molecule is polar or nonpolar, and justify ...

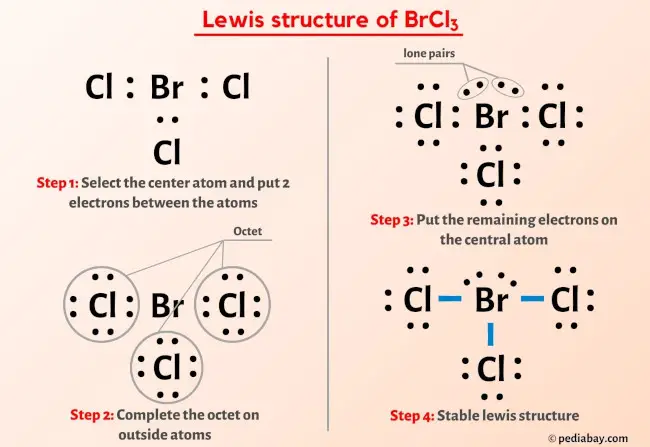
Draw the Lewis structure for BrCl3 and provide the following information. a. formal charge for each atom b. total number of electron domains c. electron geometry d. molecular geometry e. polarity
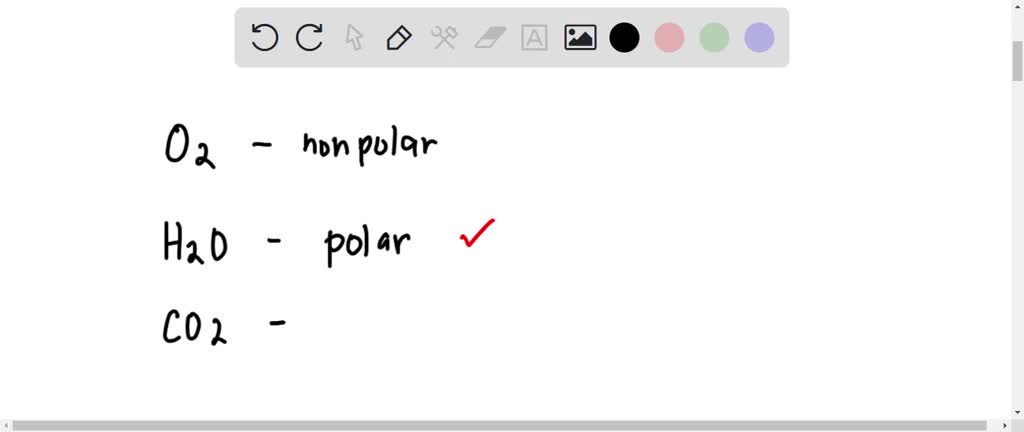
SOLVED: Are these correct? Polar chemicals Bromine trichloride (BrCl3) Bromine pentafluoride (BrF5) Sulfur dioxide (SO2) Sulfur tetrachloride (SCl4) Oxygen difluoride (OF2) Silicon tetrafluoride (SiF4) Nonpolar chemicals Beryllium chloride (BeCl2 ...