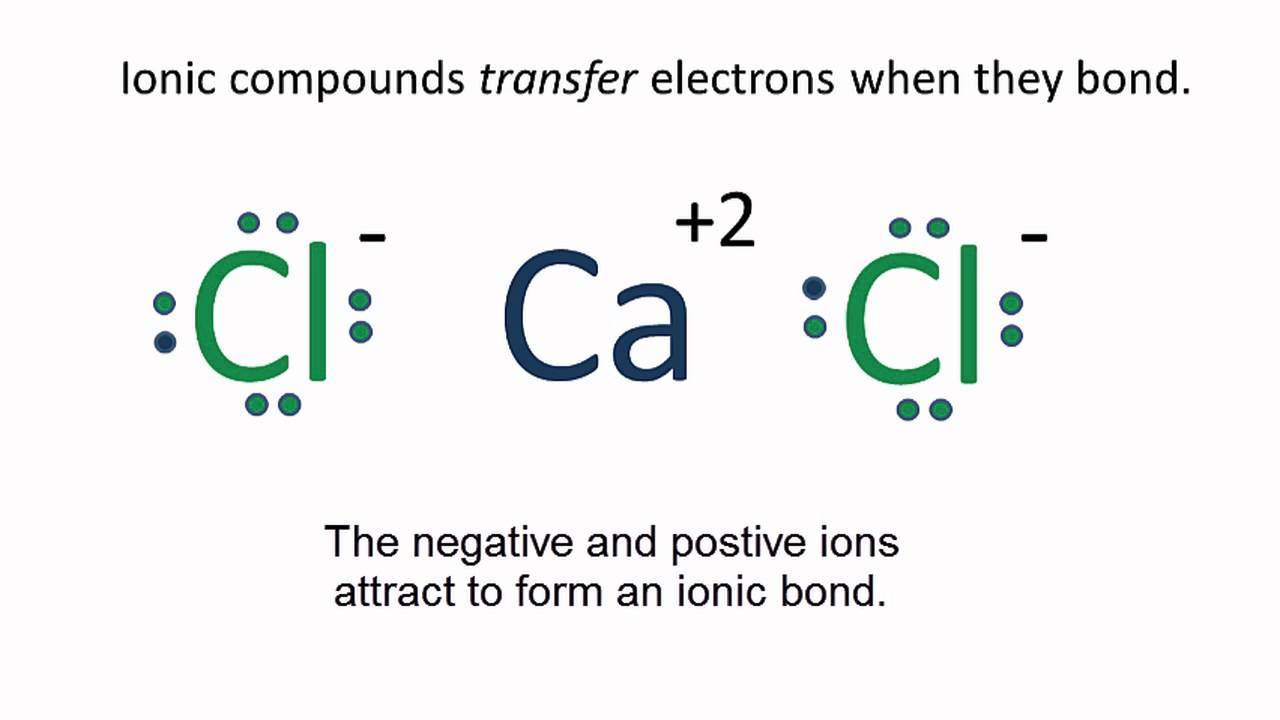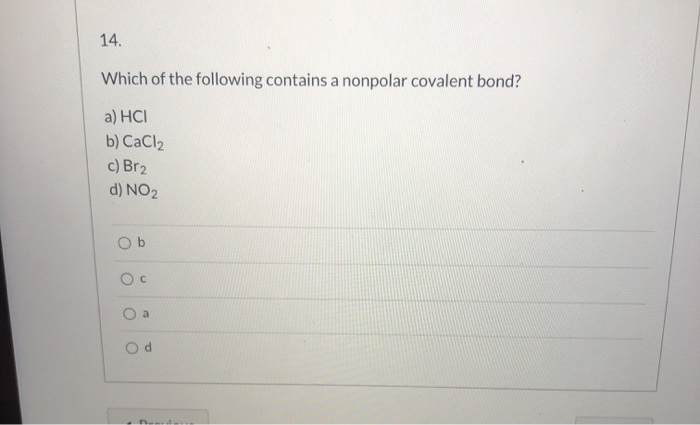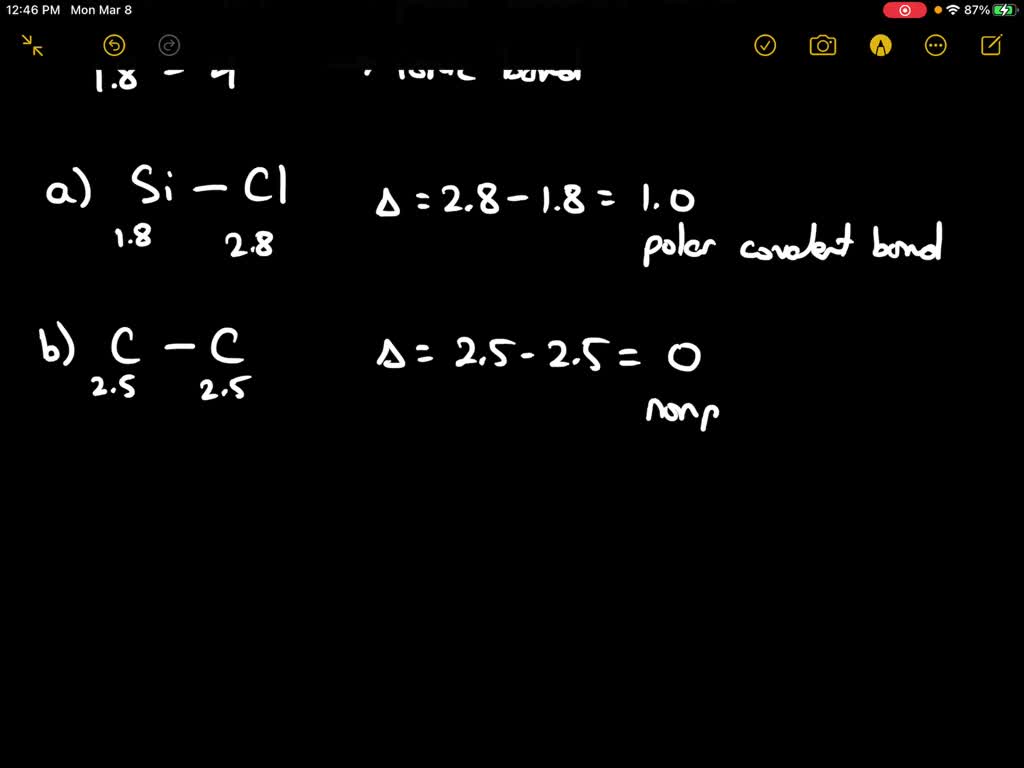
SOLVED: Calculate the electronegativity differences of the following and determine if they are largely ionic, non-polar covalent, or polar covalent? S8, CaCl2, SOCl2, NaF, CBr4, BrF, LiF and AsH3

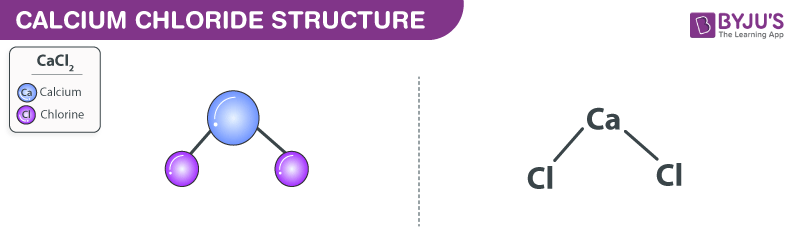
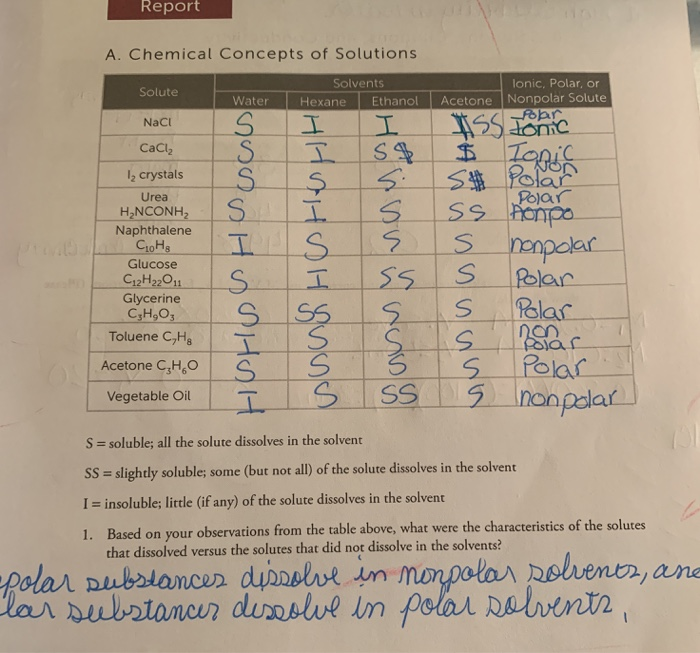
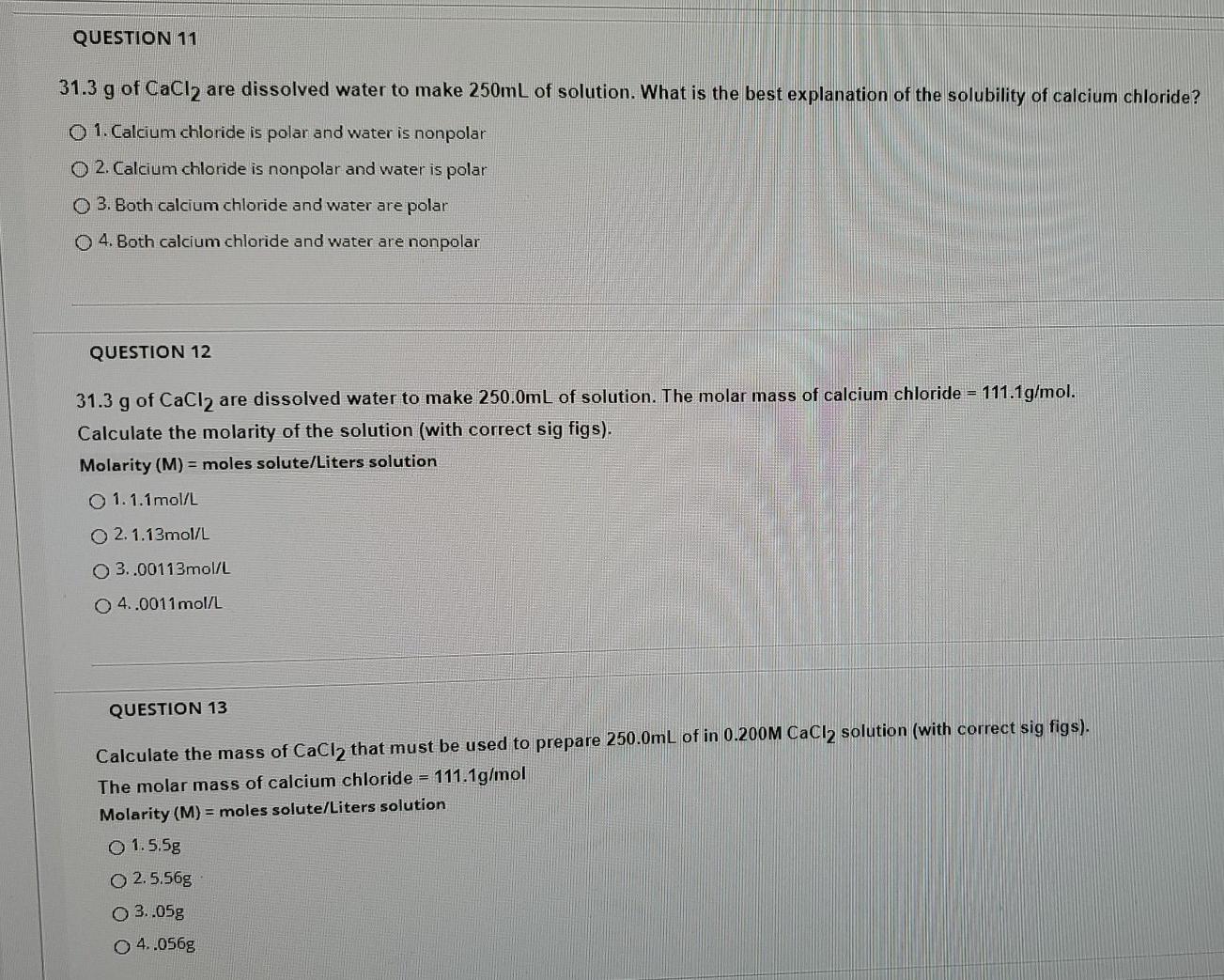
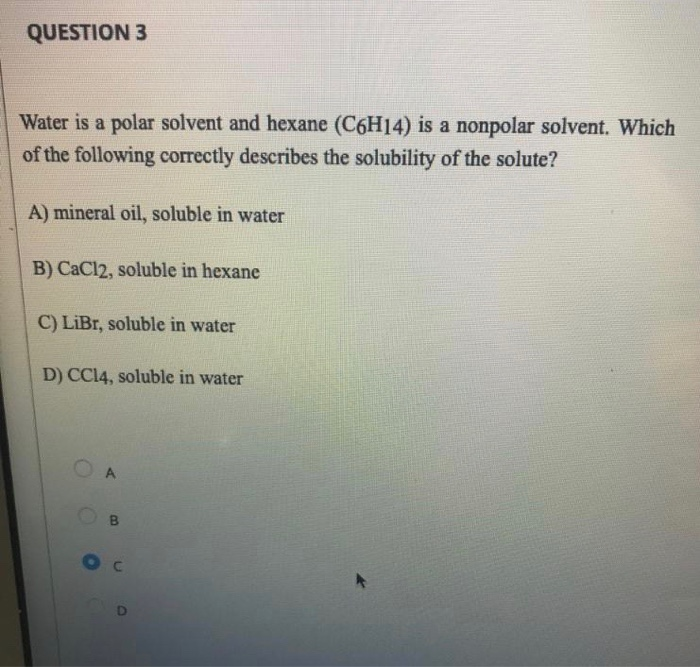
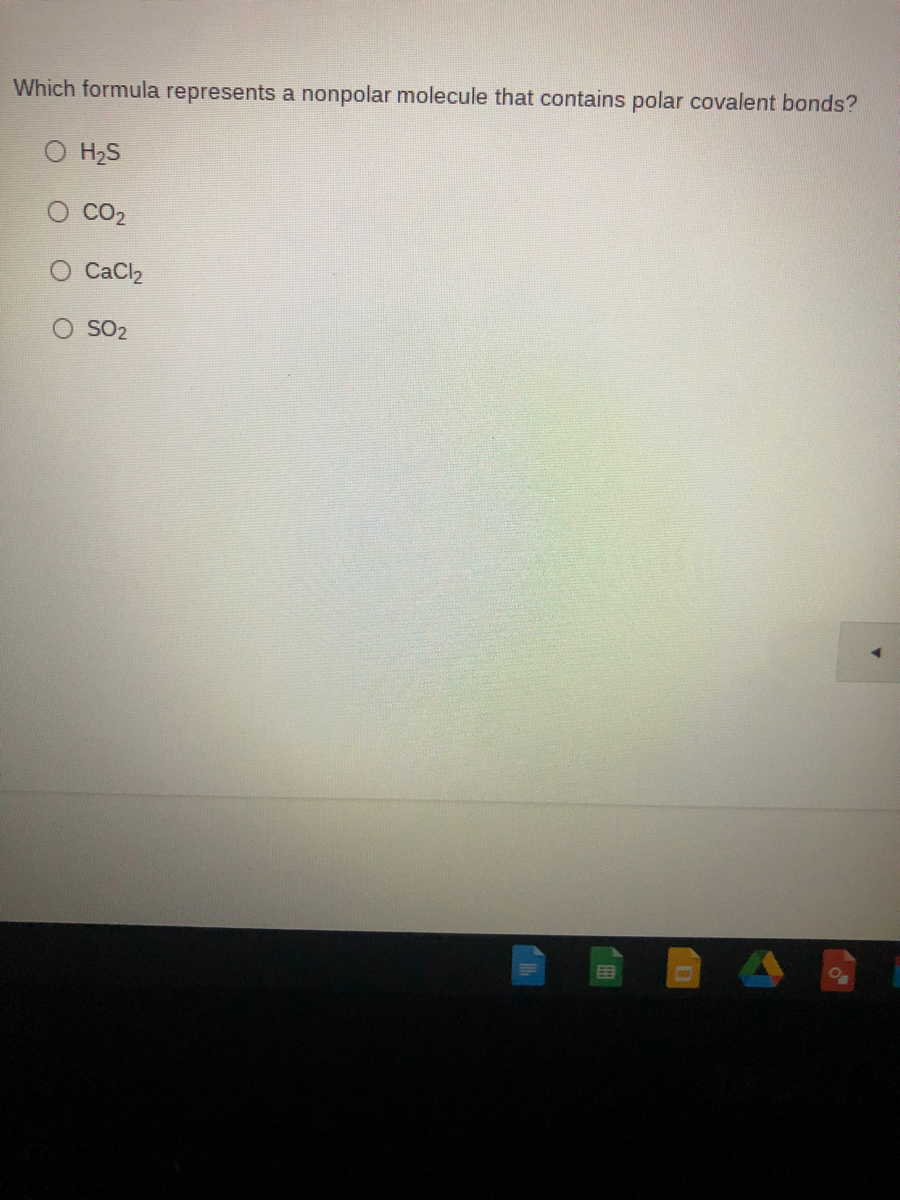
Explain whether CaCl2 is polar, ionic, or nonpolar. Then describe and explain whether it is soluble or insoluble in water. | Homework.Study.com

SOLVED: Q27 (A). Describe/draw the formation of bond in the following compounds: () CaCl2 (2 Marks) (ii) K2S (2 Marks) (iii) PCIs (1 Mark) Q27 (B) . Compare between ionic compounds with