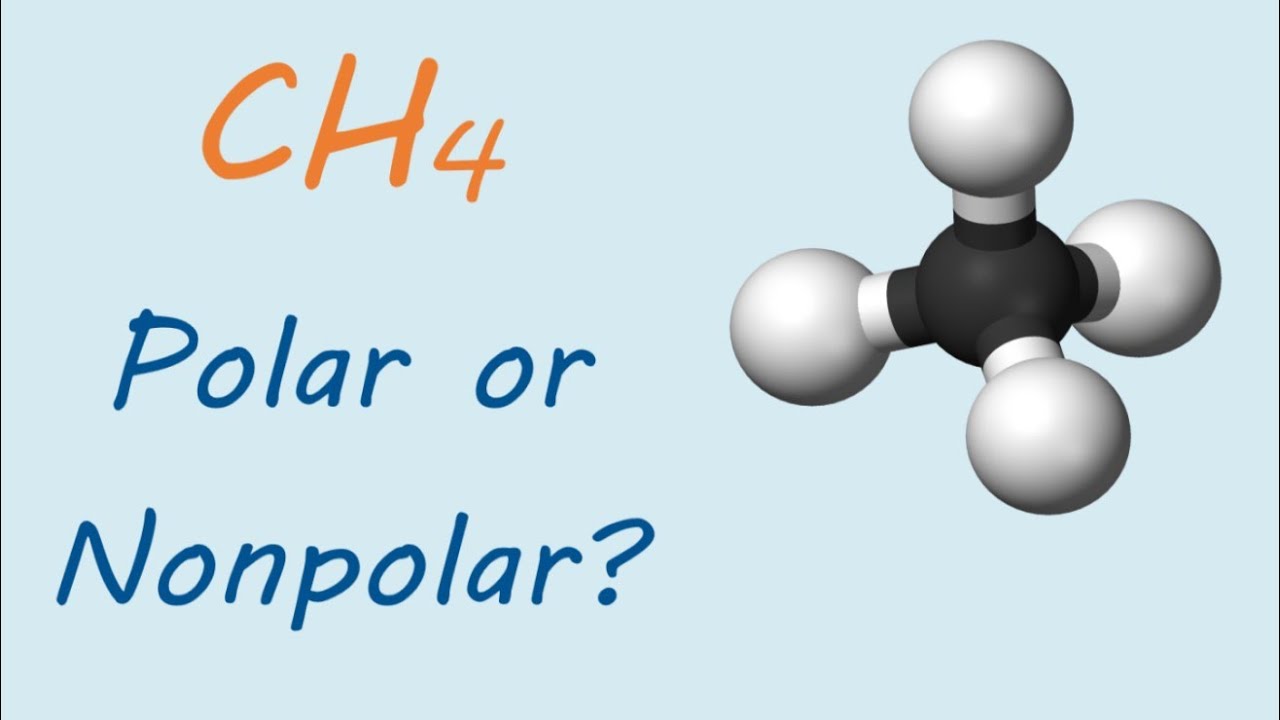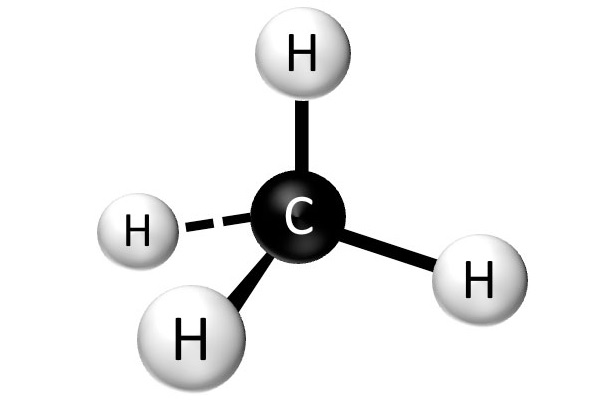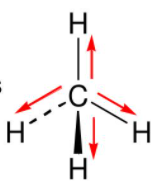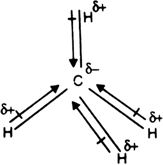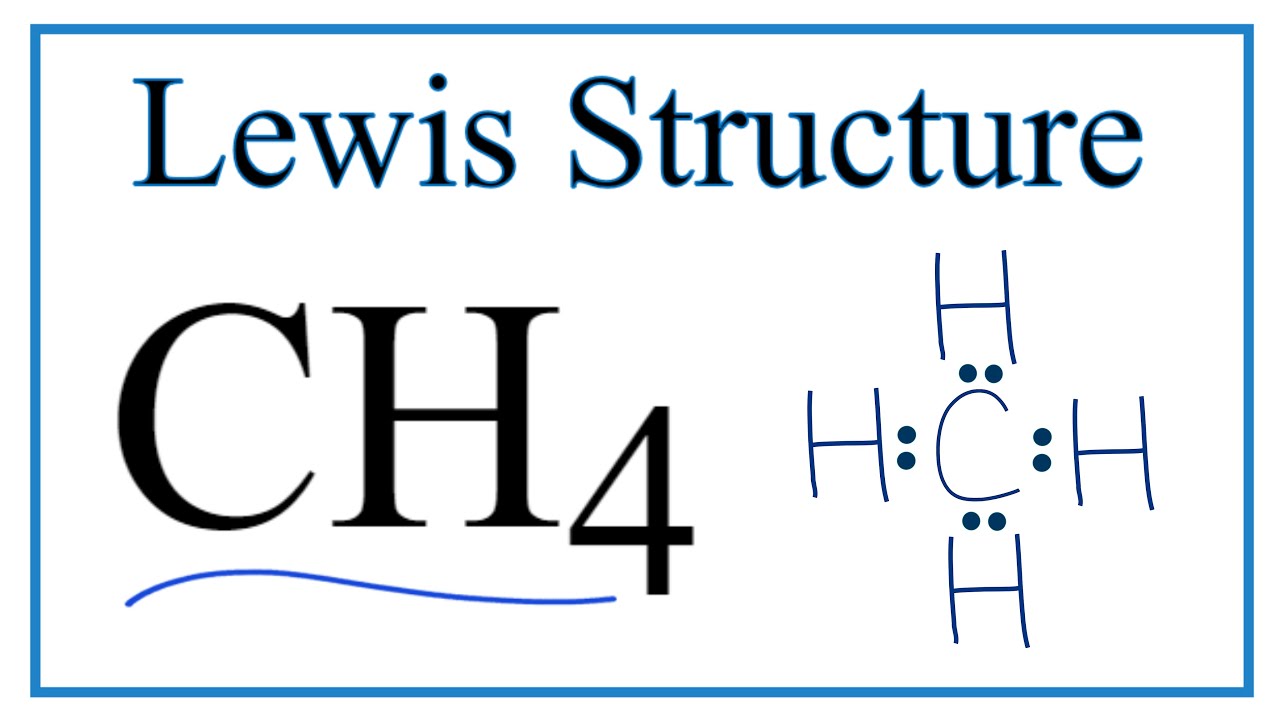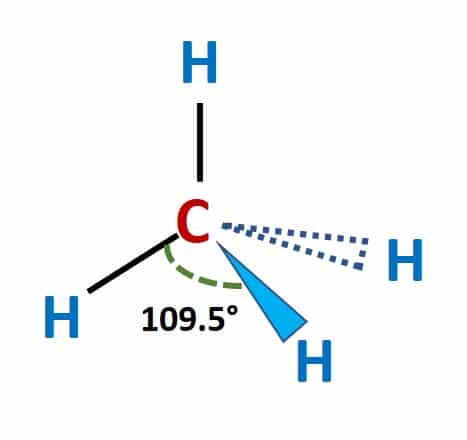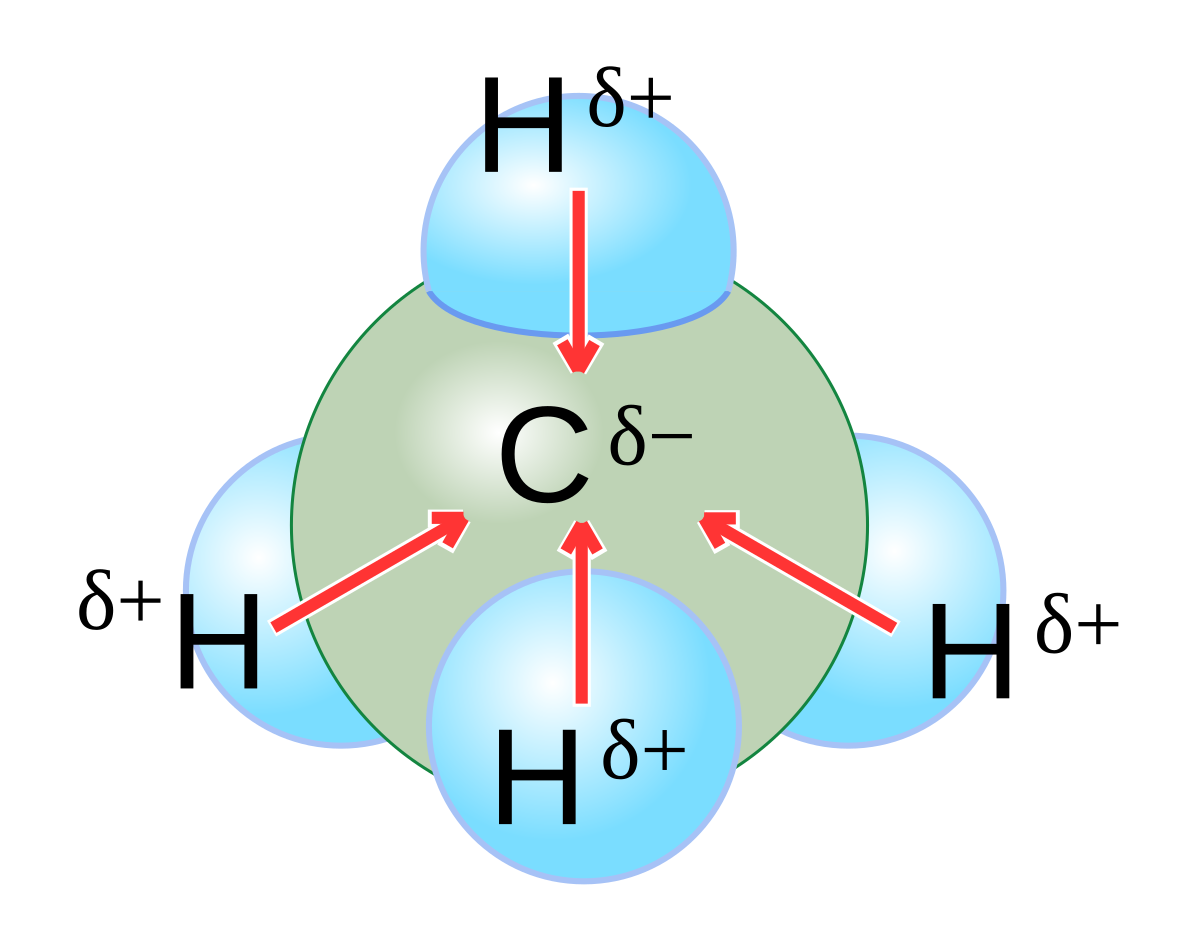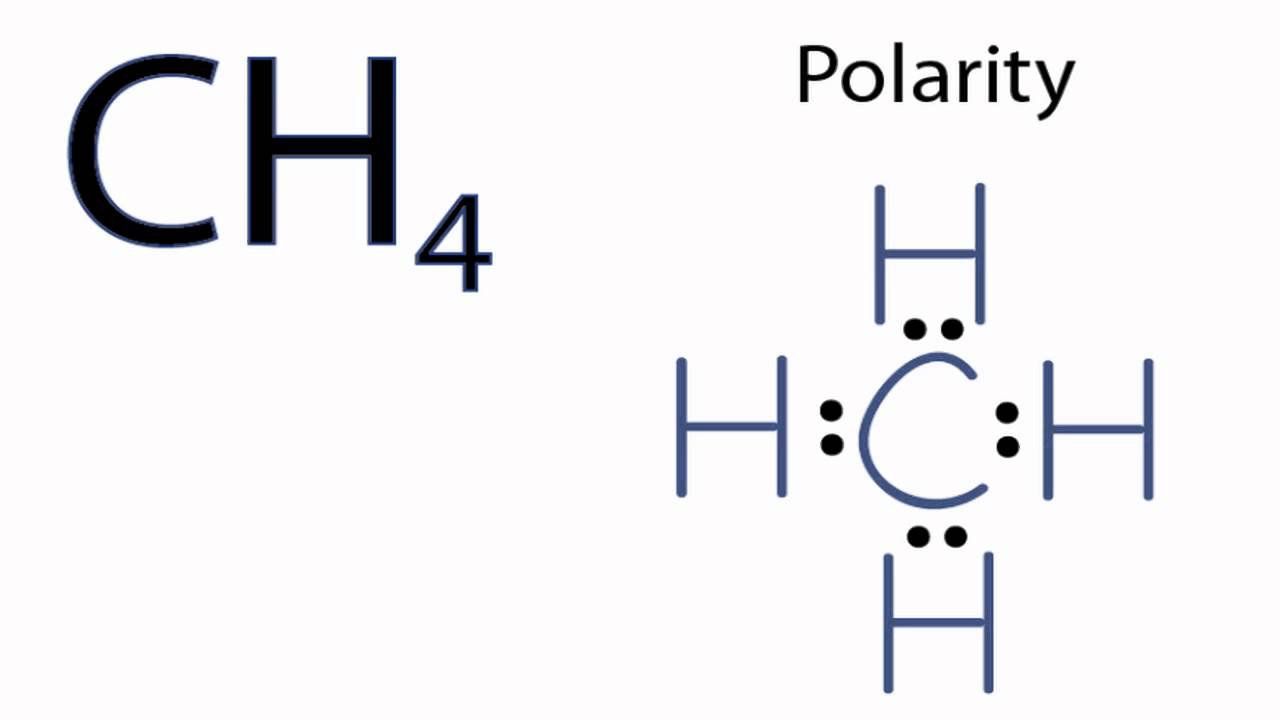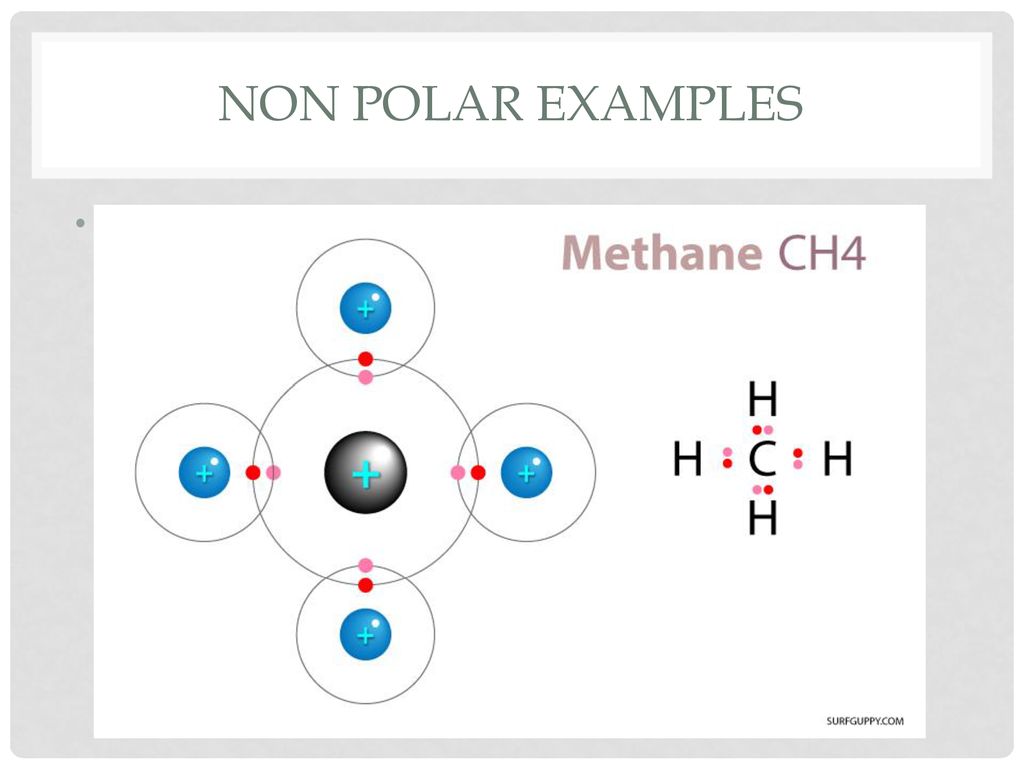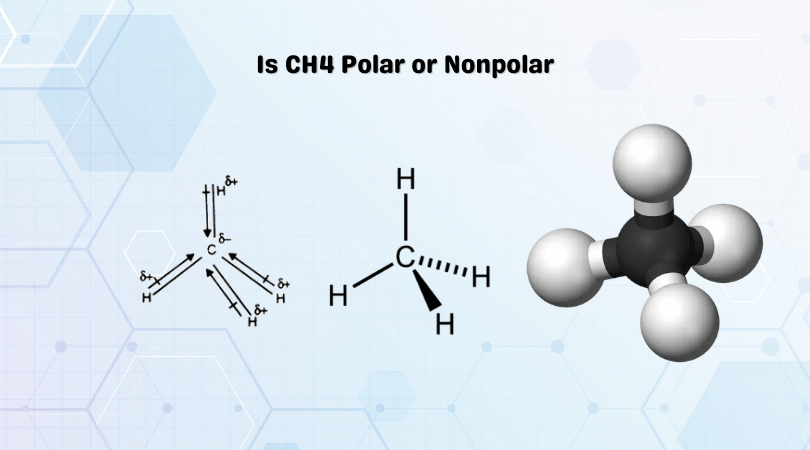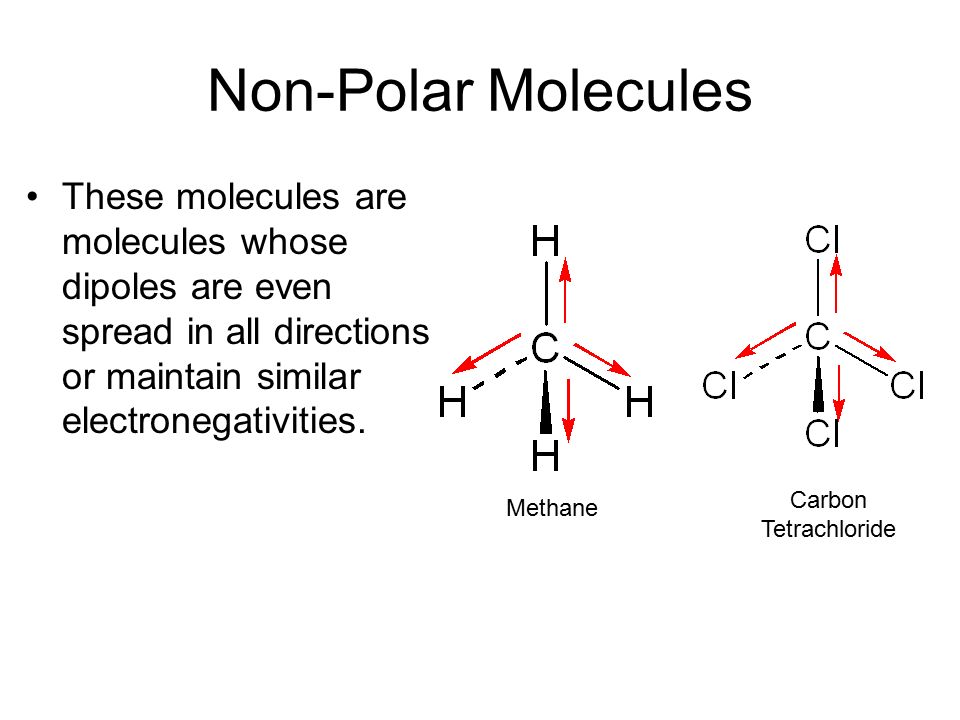
Chapter 2 Polar Covalent Bonds: Acid and Bases. Chapter 2 - Definitions Polar Covalent Bonds – electrons are even distributed between two atoms in a molecule. - ppt download

Research two common, polar molecules and two common nonpolar molecules. Draw their molecular structure and explain how the structure makes each molecule polar or non-polar | Homework.Study.com

Brianna Bibel on Twitter: "POLAR molecules, like water 💧 have *permanent* DIPOLES 👉 parts are partly ➕ & parts are partly ➖ bc they don't share ➖⚡️ electrons (e⁻) evenly 🐷 3/ https://t.co/aPZFUwBM1G" / Twitter