SOLVED: Question Select multiple answers and then press Answer. When you compare 2-propane-diol and 1-propanol, which of the following is not true 2- propane-diol is more polar 1-propanol has more number of carbon

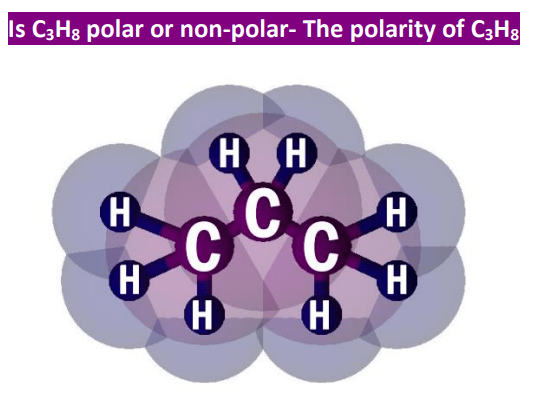
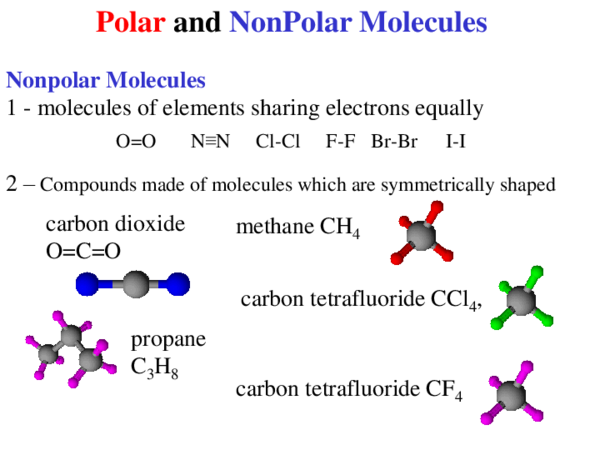
Which of the following best explains why the bond in propane (C3H8) are non polar covalent - Brainly.com